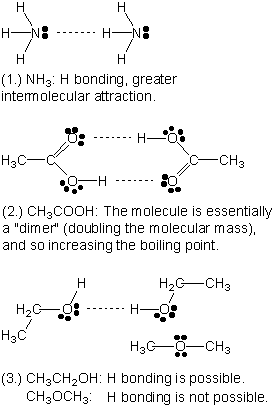
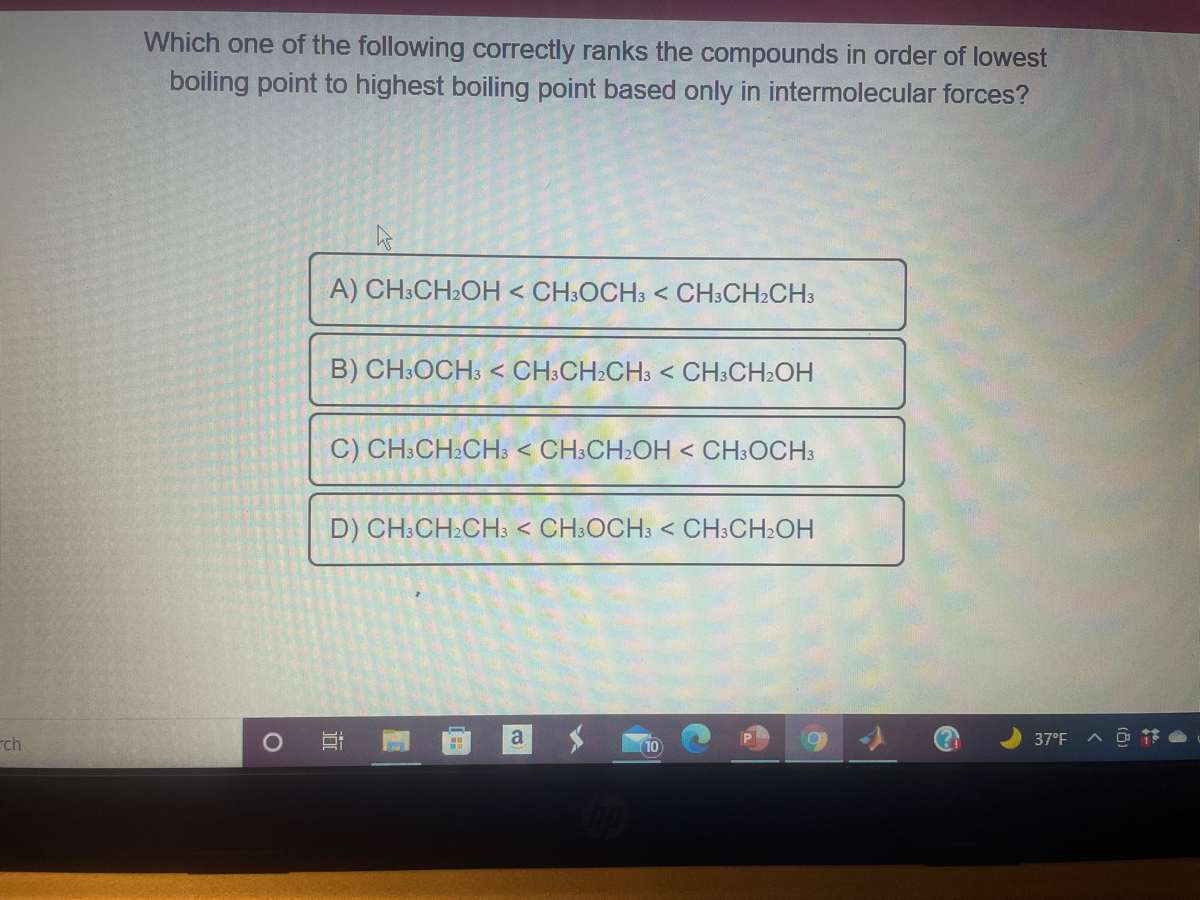
Homework 6 - 1. Intermolecular Forces T/F Decide if the following statements regarding Intermolecular forces are True or False. True: NH3 will have a | Course Hero

5 - 9701_w19_qp_11 : Intermolecular Forces, Hydrogen Bonds, Permanent Dipoles, Van der Waals - YouTube
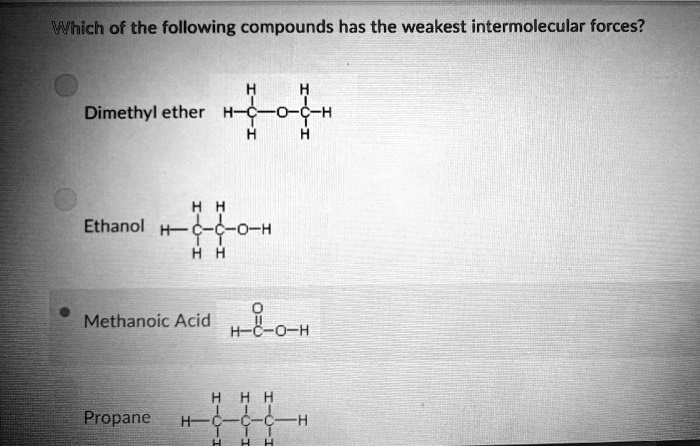
SOLVED: Which of the following compounds has the weakest intermolecular forces? Dimethyl ether C-H Ethanol H 0-H Methanoic Acid H-C-0-H Propane
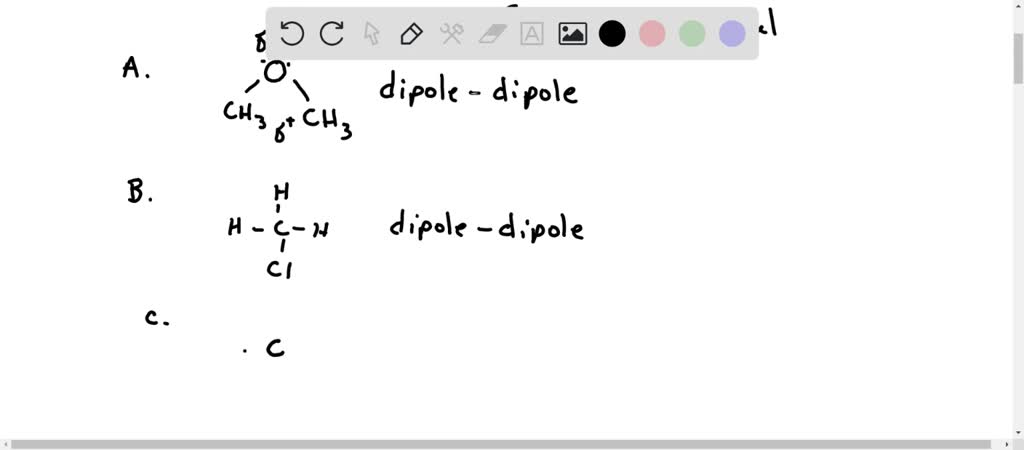
SOLVED: Identify the compound that does NOT have dipole-dipole forces as its strongest force. A. CH3OCH3 B. CH3Cl C. CH2I2 D. CO2 E. CCI3

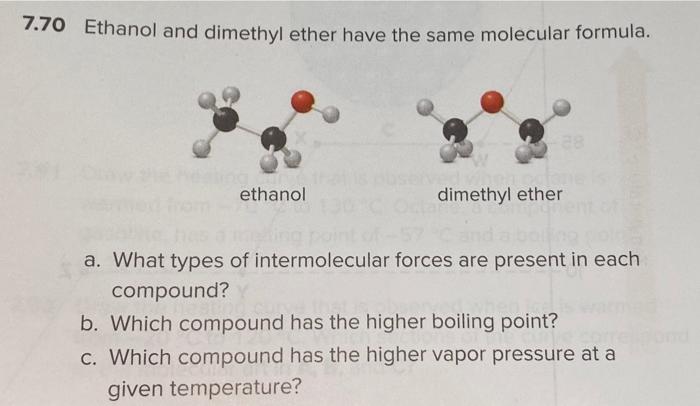
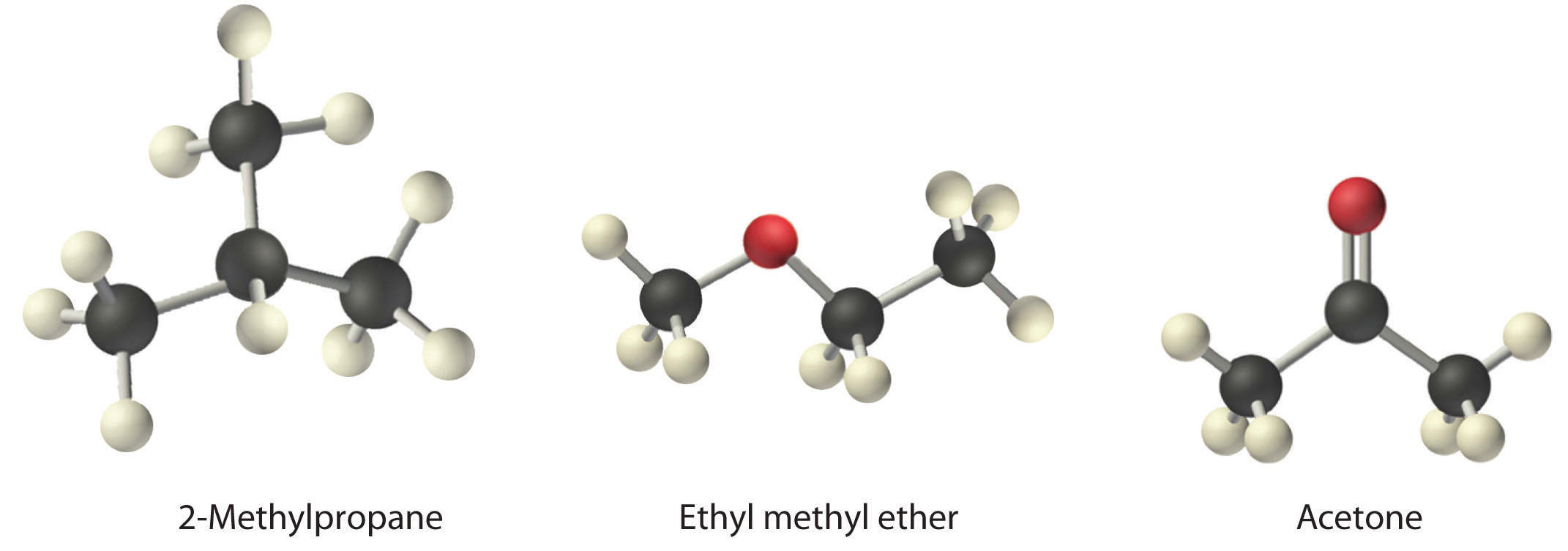
Ethyl alcohol and dimethyl ether have the same composition by mass (52% carbon, 13% hydrogen, and 35% oxygen), yet the two have different melting points, boiling points, and solubilities in water. Explain

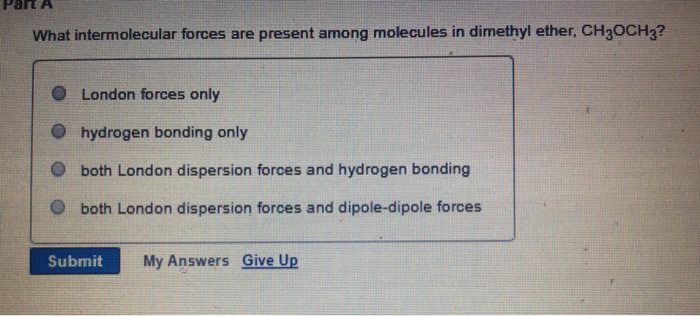
SOLVED: What is the strongest type of intermolecular force between solute and solvvent in a solution of CH3OCH3 in CsH6: 0 ion-dipole 0 ion-induced dipole dipole-dipole dipole-induced dipole