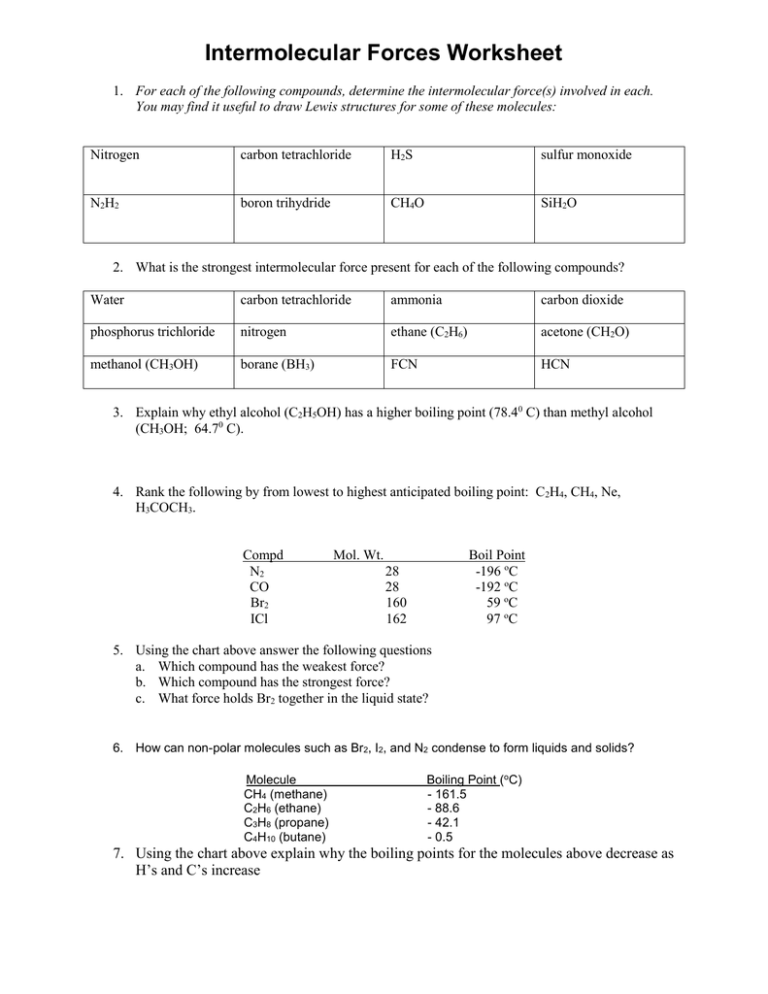
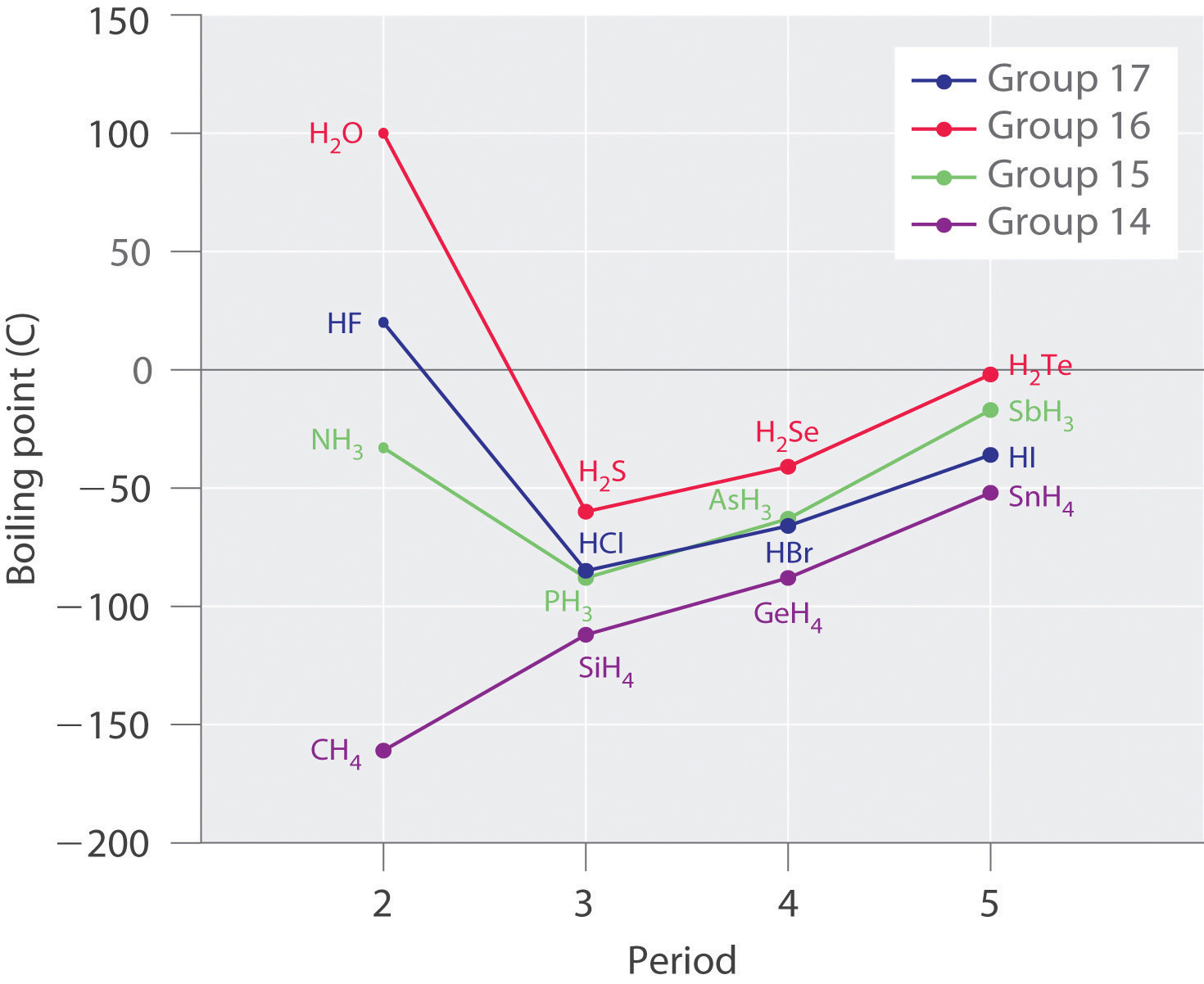
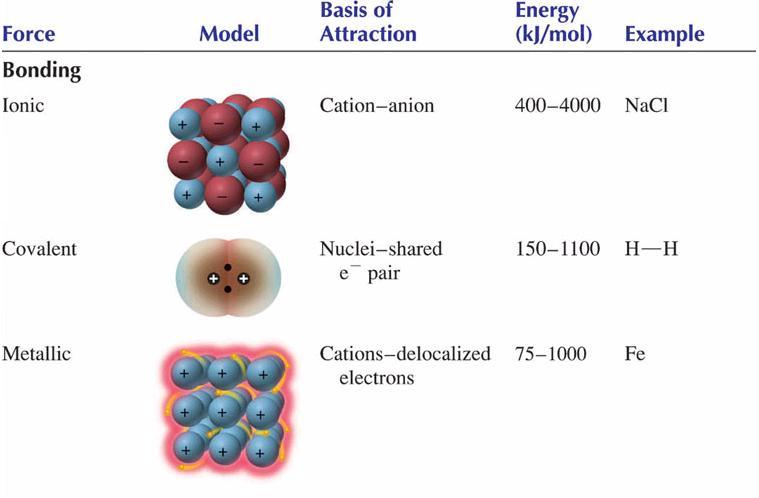
SOLVED: 3) We learned in General Chemistry the for compounds of similar size (similar molar mass) , the boiling point can be predicted based on the strength ofthe intermolecular forces (IMF's) present;

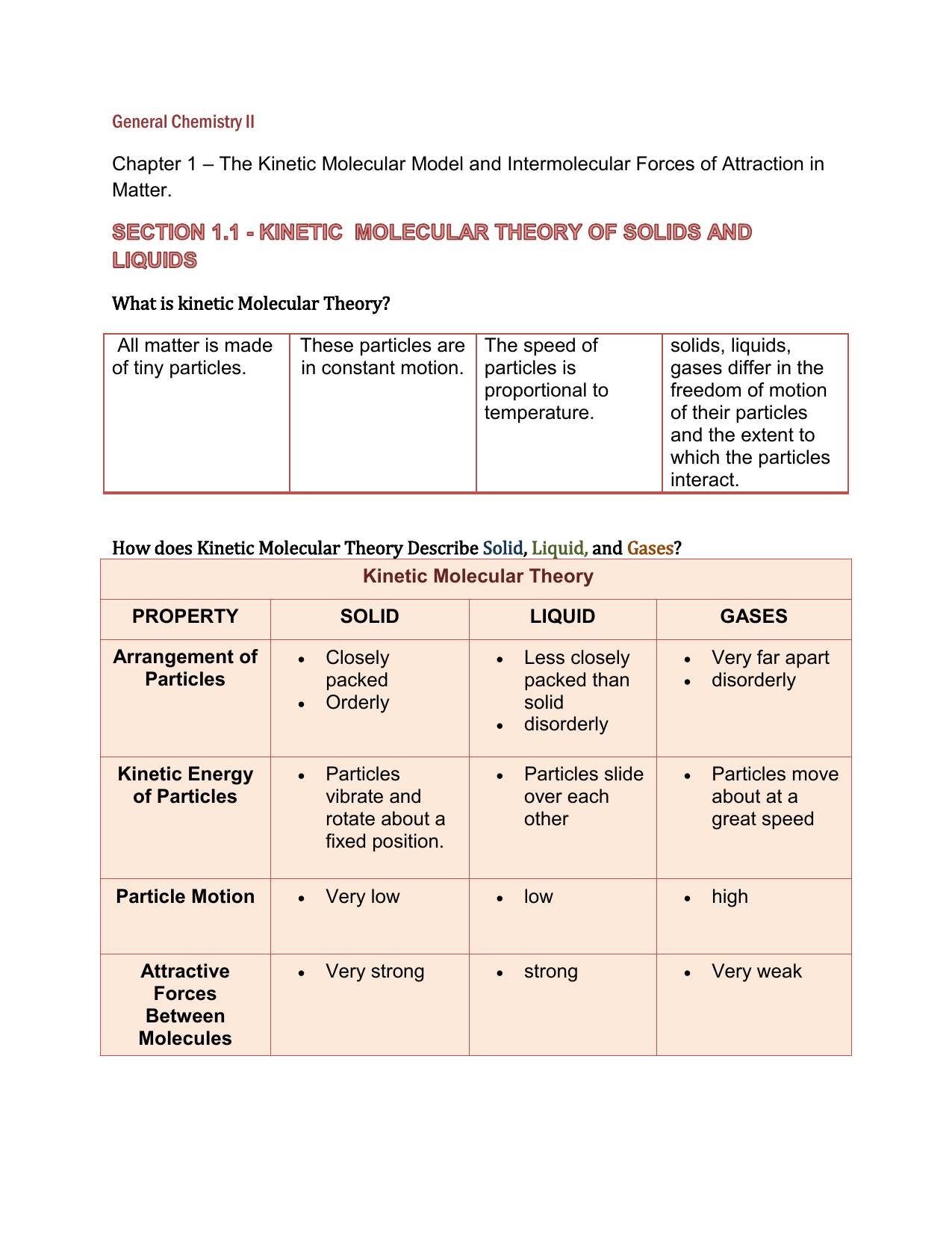
General Chemistry 2 - Chapter 1: The Kinetic Molecular Model and Intermolecular Forces of Attraction in Matter

General Chemistry 2 - Chapter 1: The Kinetic Molecular Model and Intermolecular Forces of Attraction in Matter

GenChem 2- Module 2.pdf - General Chemistry 2 Quarter 1- Module 2 Types of Intermolecular Forces General Chemistry 2 SHS- STEM Quarter 1 – Module 2: | Course Hero

SOLUTION: General chemistry intermolecular forces of attraction imfa lecture notes complete - Studypool
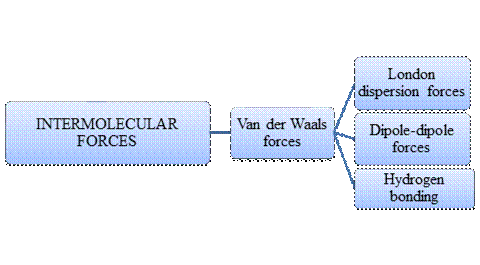
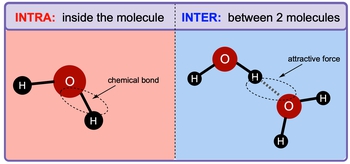
The molecular substances have moderately low melting point. This is to be explained. Concept introduction: Intermolecular forces are termed as the forces acting “between molecules” that is components of a substance. Intramolecular