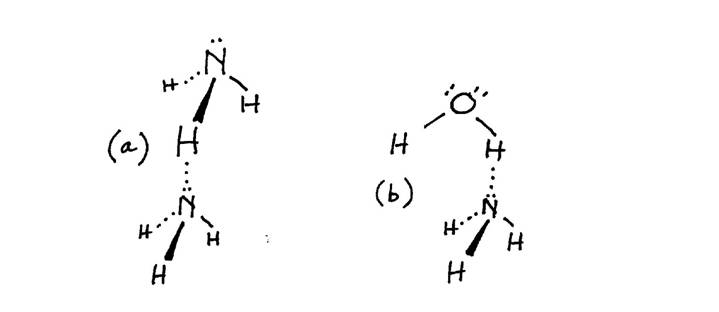
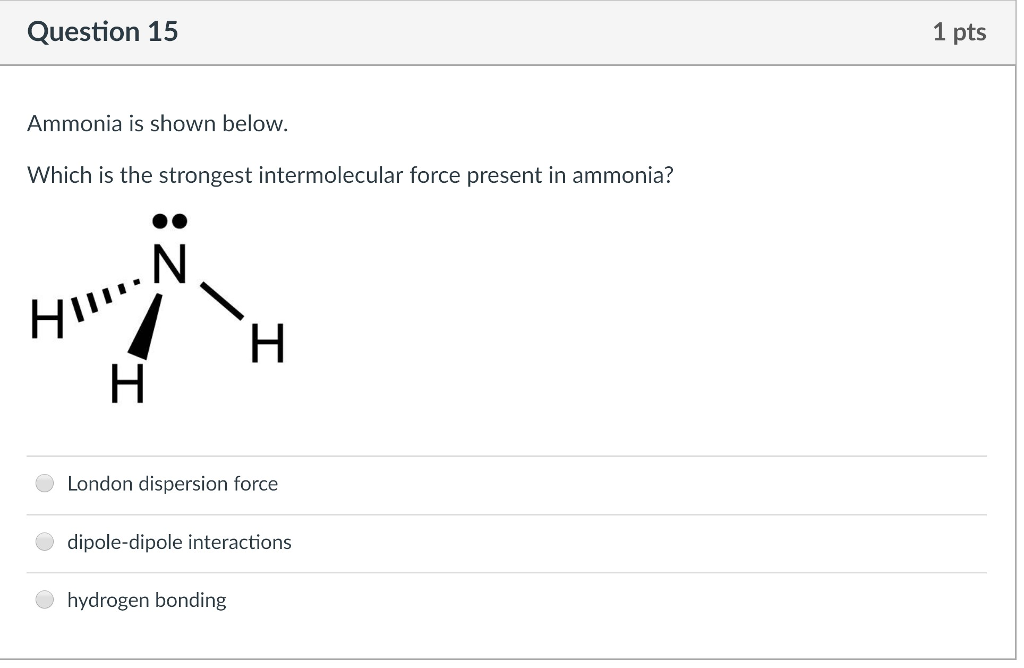
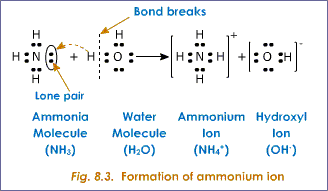
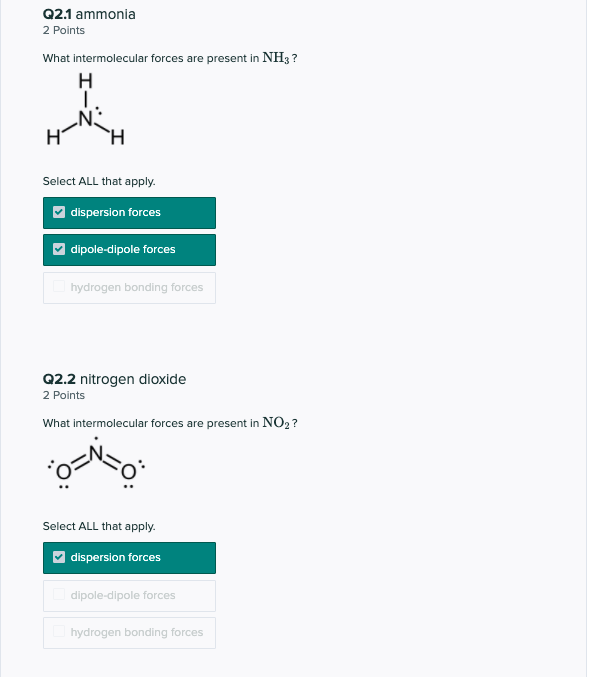
SOLVED: A Lewis structure for ammonia Is shown below: HFN-H H a. List all of the intermolecular forces present in ammonia. (4 pts) b. If NH3" is produced by reacting nitrogen gas (
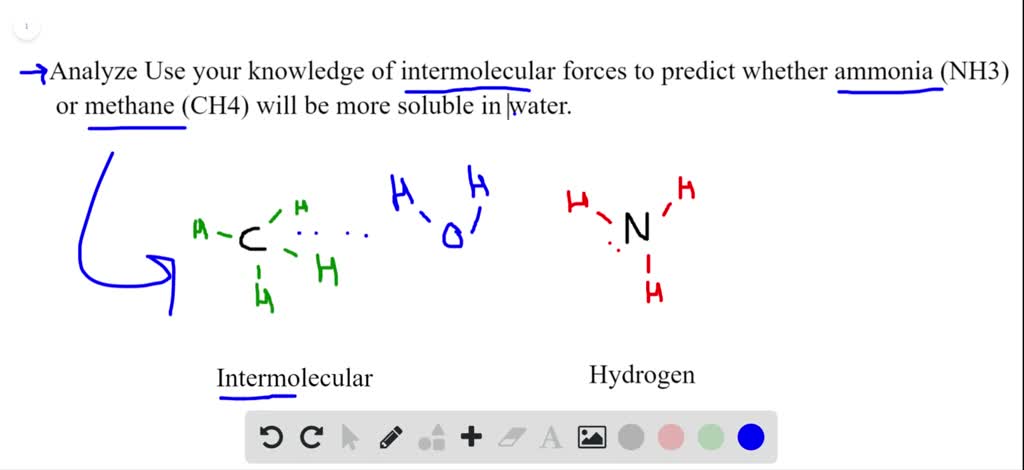
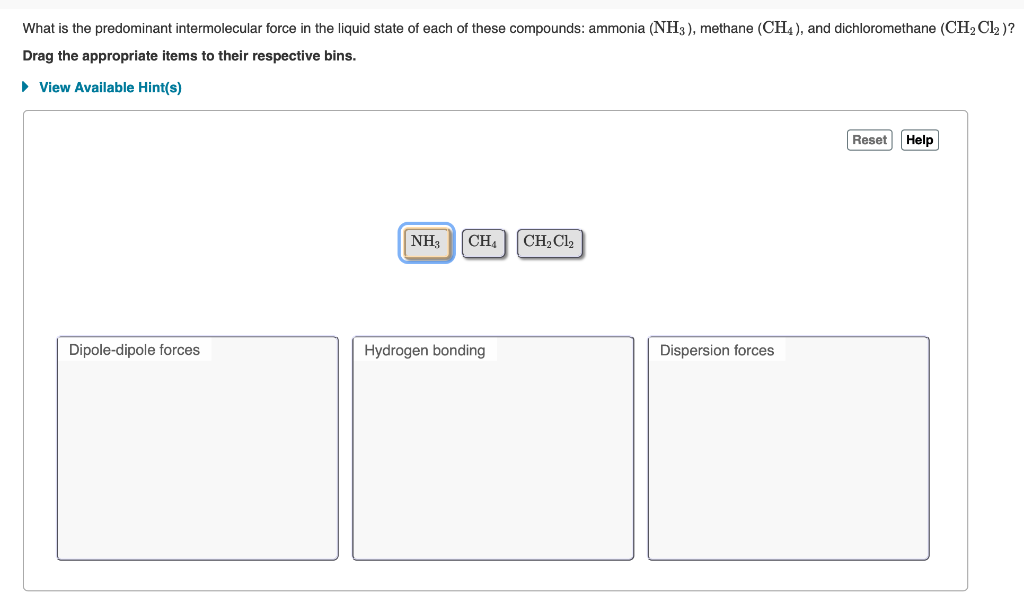
SOLVED:Analyze Use your knowledge of intermolecular forces to predict whether ammonia (NH 3 ) or methane (CH4) will be more soluble in water.

NH3, PF3, and SF6 are all gases at room temperature. What intermolecular forces are present in liquid samples of each compound? | Homework.Study.com